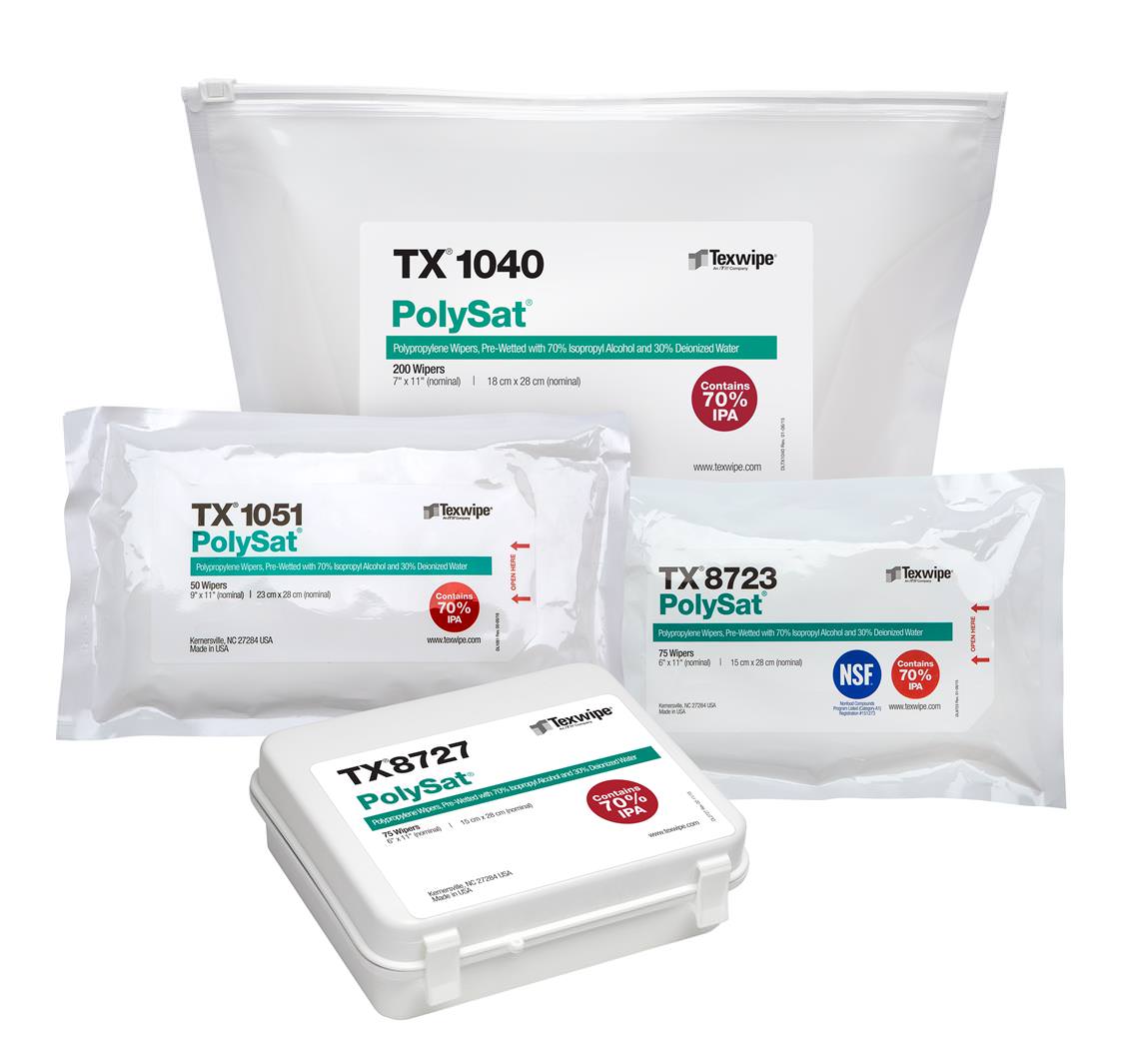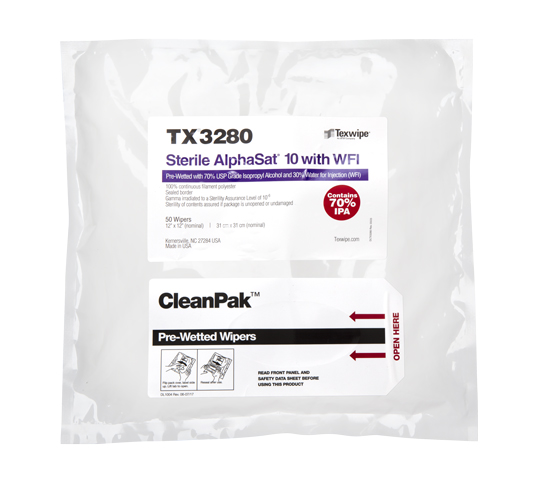
Pharma / Biologics / Lab
Texwipe® offers a full range of contamination control products for pharmaceutical, biotechnology manufacturing and laboratories. Our team of professional consultants can help you select from a wide range of product options to suit your specific application and process requirements.